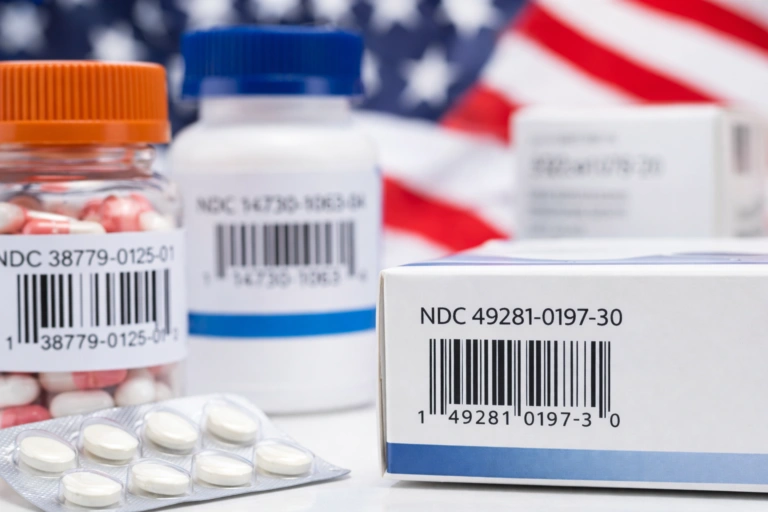
Pharma serialization solutions can revolutionize the way pharmaceutical companies sell and develop their products. By being able to identify every item along the drug supply chain, companies can guarantee that their customers are receiving high-quality products and keep them safe from counterfeit wares and imitations. Furthermore, companies can use data gathered by serialization solutions to modernize and revamp their operations.
What is serialization in pharma packaging?
Pharmaceutical product serialization is the process of implementing a unique identification code on every product along a pharmaceutical supply chain. Using serialization codes, companies trace the exact location of all their stock, regardless of geographical location.
How does serialization work?
The serialization process traces every individual product from the moment it leaves the manufacturer until it is acquired by customers. The unique code implemented on every product includes information about where it was manufactured, its production batch, expiration date, and much more.
Costs
Just like acquiring any new workplace tool or asset, there are a series of costs related to implementing serialization on a pharmaceutical supply chain. Deployment of a pharma supply chain requires companies to invest in software solutions, as well as the hardware needed for the digital systems to function.
Information about serialized products needs to be hosted inside a database so it can be accessed. If a business is taking its first steps towards digitalization, then it will also have to build the digital system infrastructure that will host the data from supply chain serialization. Nonetheless, if a company already possesses data management tools, then serialization solutions may be able to integrate with the existing systems.
Resource allocation
There is no global standard for serialization in the pharmaceutical industry. Companies looking forward to expanding their operations at an international scale must consider the particular requirements of every market they enter. Products being sold in particular countries may be subject to laws that regulate their sale and use, which could end up costing an unaware pharmaceutical company thousands in fines.
A pharmaceutical company must be ready to allocate time and resources to ensure the integrity of its supply chain. Serialization requires thorough planning, not only for the back-end of the system but for the intricacies of international trade. Proper implementation of product serialization also requires the workforce of a company to be versed in how the system works, so training may also be necessary.
Partnerships
One of the most crucial pharmaceutical serialization requirements is the formation of strong partnerships. When a company implements product serialization, it is putting the cohesion of its supply chain on third parties.
Working with dishonest partners can be devastating, as it can lead not only to loss of profits and reputation but also to legal repercussions. This rings particularly true when working with sensitive stock such as prescription drug supply chains and other vital pharma products. Therefore, it is in a company’s best interest to work with people they can trust.
Pharmaceutical manufacturers and companies work best when partnering up with experts in the field of serialization technology. Nubinno is able to meet the specific needs of any supply chain. Pharmaceutical serialization is easy to accomplish when working with an experienced team. Experts at Nubinno can deploy serialization solutions at a global scale, regardless of a company’s business type or model.
Solutions for partnered services
Automatized processes
By nature of being a digital-based solution, supply chain serialization can drastically benefit from the use of automation. By integrating automatic software tools, a company can save time and resources that would otherwise be spent manually tagging and monitoring stock.
At a simple glance, it may appear that serialization will make the manufacturing process slower. After all, unique codes must be created and assigned to every piece of merchandise, and those codes must be properly labeled on all products, cases, and pallet packaging. The man-hours required for such an endeavor could mean having to hire staff only for this set of tasks.
However, these processes can be executed instantaneously by a machine. Delegating mundane assignments to a solution that will produce immediate results is extremely cost-effective and ensures workers and management have the time to take value-added tasks.
Data management
Data is an extremely valued commodity. The big data analytics market was reportedly worth over $200 billion in 2020, and estimates say that number could see a two-fold increase or even higher before the end of the decade (source). Some of the fastest growing companies of the last few years, such as Google, Amazon, and Meta, rely heavily on the usage and transfer of data.
Nonetheless, without proper management and analysis tools, data is just code building up space in a database. Effective supply chain serialization requires a company to deploy efficient data management and reporting software. Access to this type of software may be supplied by partners. Nubinno, for instance, works alongside large Cloud Analytics providers to give their partners the best product management services.
Data exchange
If a company is running pharma supply chain operations, then it is bound to send and receive data from a wide variety of sources. Product serialization requires the constant monitoring of merchandise, and that is achieved by scanning stock at every location. For example, if you’re shipping from Japan to the US, data will be created when a product comes out of manufacturing, when it leaves Japan, when it arrives in the US, and so on.
However, just like someone can’t travel to another country and expect to understand the language, software apps in different locations may not communicate efficiently. Data exchange is the process of transforming the structure of data so it can be accurately portrayed by different applications. If your company is on the lookout for product serialization solutions, then it must look for providers who are data experts.
Regulatory requirements
Entering new markets must always be done so with careful attention to all laws and regulations. Overcoming serialization regulations can be a challenging struggle, not only due to the sensitivity of the subject but also because of the constant changes enacted by governments to the existing laws.
Current regulations regarding the trade of pharmaceutical products can vary widely between countries. The US Food and Drugs Administration (FDA), for instance, is a governmental body with more than 2000 internal reviewers ready to swiftly process new drug applications, while China’s National Medical Products Administration (NMPA) is a relatively new regulatory body, which performs at a slower pace than its western counterparts.
Pharmaceutical serialization requirements
Since legal pharmaceutical serialization requirements vary between countries and economic regions, a company can’t adapt a one-fits-all approach to this process. However, legislators tend to use the guidelines of the GS1 organization when placing regulations on product serialization. The GS1 organization is an international non-profit devoted to creating global standards for pharmaceutical track and trace practices.
The GS1 advises pharmaceutical companies and regulatory bodies to implement the following information on serialization codes:
- Global Location Number (GLN) and Global Trade Item Number (GTIN): These numbers should be available to those who register with their national GS1 association. The GLN will work as an identifier of a company’s operating address, while the GTIN is a unique code that identifies products at a global scale.
- Unique Serial Number: Every product should contain either a numeric or alphanumeric character sequence of up to 20 characters. Serial codes must be unique for every GTIN.
- Batch or Lot information: Just as the product itself, information about the batch it was transferred in should also be provided.
- Expiration Date: Proper information about a product’s expiry date. All expiration date information should be accurate and easy to access.
More information on the GS1 stipulations can be found in the GS1 DataMatrix Guideline.
More than 110 countries have local GS1 member organizations and practices suggested by the GS1 facilitate the execution of 6 billion daily transactions in 150 countries (source). Even so, a company can’t suppose a country has implemented the GS1 guidelines verbatim, so performing the appropriate research and growing a business development network are recommendations all pharma companies should follow.
As more countries implement serialization regulations, companies must adapt their practices to fit the rules. This makes the early adoption of serialization practices a long-term value proposition, as the technology is already becoming a global standard. Egypt, for instance, began implementing the GS1 guidelines as recently as 2019 (source), and many other countries are looking to follow.
Pharma serialization services
With a main emphasis on consulting and implementation of digital serialization solutions for pharmaceutical products, the experts at Nubinno are ready to help any company expand its business to the global market.
Nubinno is comprised of a culturally diverse consultant team with the ability to communicate in more than 10 languages. Companies partnered with Nubinno operate all over the world, with projects being handled in all of the EU, the US, India, China, Brazil, South Korea, Arab countries, and more regions.
Nubinno’s world-class services can adapt to different business models. It doesn’t matter whether a business has a particular business type or if it is restricted by geographical reasons, Nubinno can devise unique strategies to ensure success.