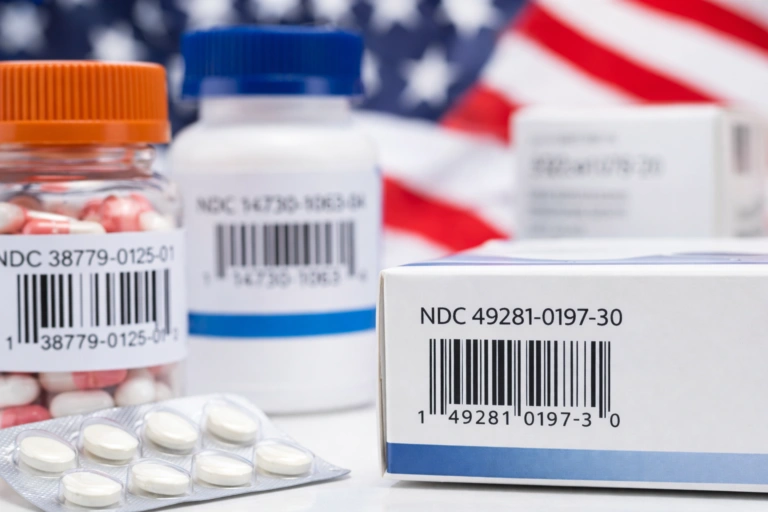
What is the 2023 DSCSA deadline?
The 2023 DSCSA deadline is the point in the future from which the FDA will require pharmaceutical companies (manufacturers, distributors, wholesalers, and dispensers) to abide by new serialization requirements. It is currently set for the 27th of November 2023, and while that may seem far away, one can never be too early when preparing for these deadlines. Case and point, there was another deadline for this same thing in 2018. However, nearly half of pharma manufacturers failed to meet it. This was not all due to tardiness, however, as problems also stemmed from unclear requirements.
That is why it is so important to try and meet the 2023 DSCSA deadline, and this article will help you. It will examine what those requirements are and help you meet them, as well as talk about the reasoning behind them.
What are the requirements for meeting the 2023 DSCSA deadline?
According to contactpharma.com, there are three main requirements that pharma manufacturers, distributors, wholesalers, and dispensers need to meet by this deadline to continue along the supply chain:
- Interoperable exchange: Authorized trading partners are required to share transaction information (TI) and transaction statements (TS) securely and electronically. The TI should include the product identifier (PI) at the individual package level.
- Interoperable verification: Partners must interoperably and electronically verify the PI at the unit level. Essentially, they must confirm that the shipments they receive match the corresponding electronic data.
- Interoperable tracing: Unit-level traceability (i.e., serialization) is required so that each individual product can be traced throughout the supply chain. A PI includes the product’s standardized numerical identifier (SNI), an expiration date, and a lot number.
What is the purpose behind the 2023 DSCSA deadline requirements?
As with most things regarding the FDA, safety is the primary concern that is being addressed here. Despite the many improvements in serialization systems in the past two decades, counterfeit drugs still make their way through the cracks. This has unwanted side effects such as smuggling as well as wholesale medicine being coated with addictive substances that make patients come back for more even though their initial reason for taking the medicine might have disappeared. There have even been some fatalities caused by this.
As you may have noticed from the requirements, the main idea is to make the drug’s journey along the supply chain as transparent as possible. Interoperability is emphasized so that tracing and checking become simple and efficient. The large focus on communication between parts of the supply chain will further be explored as we tackle the necessary mindset and actions for meeting the 2023 DSCSA deadline.

The way(s) to meet the 2023 DSCSA deadline requirements
Now let’s talk about everything that will be necessary from pharma companies in order to meet these requirements. No doubt all of this sounds good on paper, but achieving it is another thing. For this reason, we will now explore the principles which have to be employed if this deadline is to be met.
As we have mentioned, due to a lack of clarity regarding the specifications and who was responsible for fulfilling them, over half of pharmaceutical firms missed the DSCSA’s initial deadline in 2018. The FDA’s failure to establish precise rules was a contributing factor, but poor internal communication was also part of it, and it is exactly that which we will discuss first.
1. Fixing bad communication
Bad communication happens both on the inside and outside. Meeting the 2023 DSCSA deadline will require that the FDA be more clear with their requirements, yes, but it also demands that pharmaceutical companies themselves step up their communication game. Confusion over the licensing standards set forth by the FDA—and who is responsible for addressing them—can lead to duplication of effort or the failure to finish a task entirely. Establish a company-wide plan with a location to direct issues or complaints, a clear schedule for when each action item must be accomplished, and roles and responsibilities for teams and individuals. Likewise, strengthen the communication between yourself and your trade partners. After all, the focus of the deadline requirements largely revolves around interoperability.
Good communication also implies better planning. A good plan will help you avoid the 2018 situation from repeating. Instead of seeing this plan as a list of boxes to be ticked off, you should think of it as a process of trial and error. In actuality, this entails setting aside some time before the deadline in November to resolve any problems. Furthermore, you shouldn’t conduct your planning in isolation. To make sure everyone stays on the same page, keep in constant contact with the manufacturers, distributors, and dispensers you work with along the supply chain.
2. Updating software and protocols
Have you updated your serialization protocols? Manufacturers, packagers, and wholesalers traded TI and TS on a lot-by-lot basis as of 2015. Beginning in 2023, you will need serialization down to the package level to achieve unit-level traceability. To meet the 2023 DSCSA deadline, your systems will need to be reevaluated to see if they are capable of compliance. For instance, you will now need to track each sellable unit you receive and send instead of just tracking packages at the lot level. Your business needs to be able to manage complicated product information in real time in order to do this. Employing enterprise resource planning (ERP) software, which manages these moving elements for you and keeps your company compliant, is one choice.
Keep in mind that there are always enthusiasts who perceive possibilities to use these new processes and technology for their personal gain, even if most pharmaceutical businesses do not get enthusiastic when new laws are imposed. Are you one of them? This is precisely why we created the Nubinno Accelerator service, which enables us to thoroughly examine the short and long-term goals and ambitions of our clients. This enables us to pinpoint the aspects of serialization compliance that can be improved and aligned with those of our clients.
3. Updating hardware
It is not just your software that you need to update. In the past, once departing carriers scanned a linear barcode, the product cargo was ready for shipment. Now that a different kind of barcode is used in the outgoing process, you probably need new hardware. If the 2D barcode scanners your business currently uses cannot read this new sort of barcode, look into other possibilities and make the appropriate investment. This also applies to RFID readers. Additionally, you must ensure that the necessary hardware is up to date if you are adding new software or other digital tools to your tech stack. It is also crucial to identify necessary hardware upgrades as soon as possible because buying and implementing new hardware can be a time-consuming procedure.
If you are having trouble assessing your company for these hardware gaps (as well as those related to other business aspects), then Nubinno Assessment might be for you. We can help you analyse your business in four key areas (processes, people, technology, and data) so you can know exactly what improvements need to be made and how to make them.

The importance of meeting the 2023 DSCSA deadline
The DSCSA has said that it would have no further delays in the deadline. This means that it is very important for pharmaceutical companies to take it seriously and meet the requirements. If not, there are many perks which they will be denied, the most problematic of which is probably no longer being an authorized trading partner. The DSCSA restricts authorized trading partners (ATPs) to only conducting business with other ATPs. To put it another way, all producers, wholesalers, distributors, repackagers, 3PLs, and dispensers, as well as their business associates, must be ATPs. Their access to the American pharmaceutical supply chain will be severely limited or denied if they are not authorized.
There are other reasons beyond just repercussions from the DSCSA. The requirements for meeting the 2023 deadline focus on modernizing a company’s serialization systems. Those that fail to meet them risk being left in the dust as their competition paves the way for the future. Meeting the deadline requirements is also a direct way to update and organize some outdated systems you might be operating with.
How to be sure I have met all the requirements?
Despite the DSCSA being clearer with their requirements this time, there can still be doubts regarding whether or not you met the deadline. This is because lots of little details can slip in through the cracks. One scenario you definitely do not want is for the 2023 DSCSA deadline to come up and for you to be sure that you have fulfilled all of the requirements, just to discover that there was an oversight somewhere which does not make your organization legible for meeting the deadline.
The most reliable way to make sure this does not happen is to have an audit. Hire a third party to go over your serialization systems and check if everything is ready for the deadline. If so, then you can rest assured and wait for the deadline to come with no fear. If not, they can advise you on the next steps you need to take to meet the deadline. It is a win-win situation that will ensure you meet those requirements! If you have any doubts either way, it is a great option.

Final thoughts and some additional help
We hope that this article has been a helpful and informative introduction to just what the 2023 DSCSA deadline is and how to meet the requirements for it. Pharma businesses will need to enhance their procedures to boost visibility into the pharmaceutical supply chain as long as the problem of counterfeit pharmaceuticals exists. Although overhauling operations twice in a decade is no easy undertaking, the benefits to your business and public health are enormous. You may establish the groundwork for ongoing development in 2023 and beyond by agreeing to satisfy the phase two requirements.
Looking at these requirements, however, it can be a bit intimidating to consider how to meet them. Do not worry, though, for they most likely only seem complicated. If you know what you are doing, the practical aspect will not be that difficult to take care of. On top of the services already mentioned in this blog post, we at Nubinno have lots of experience in helping pharmaceutical companies with their serialization projects. The 2023 DSCSA deadline requirements are simply more of the same—update, innovate, connect, and organize. For this reason, if you feel like you need some extra help with figuring out how to meet these requirements, feel free to reach out to us and we will see how we can help you with a more hands-on approach.